Groups and chemistry: a nice application of Character theory
Introduction
Group theory is closely related to the study of symmetries, be they symmetries of abstract mathematical
objects, or of more concrete ones such as molecules. Explicitly, group theory can be applied to
infrared (IR) spectroscopy in order to predict the presence of absorption bands:
If a radiation excites the molecules from the ground state into vibration, it is absorbed.
Suppose we have a list of candidate molecules for an unknown sample (which we suppose to be
composed of only one molecule).
Since the frequencies absorbed by a molecule are characteristic of its geometrical structure,
by knowing the geometries of the candidates, we can calculate the expected number of absorption bands,
which may eliminate non corresponding candidates when the actual IR spectrum is determined experimentally.
Background on Character theory
I don't provide here a course of character theory from scratch, but if you do have notions of group theory and algebra, the basic definitions and properties needed to understand this article are available here.The general Method
- First, determine the symmetry group `G\leqO_3` of the molecule points `P` in `\mathbb{R^3}`, and compute its character table and `\chi_0` its standard character associated to the permutation of `P`.
- We consider `\Gamma:={f:P\mapsto\mathbb{R^3}}` the set of infinitesimal displacements of `P`.
The action of `G` on `\Gamma` is defined as :`\forall f:P\mapsto\mathbb{R^3}`, `\forall g\in\G`, `forall x\inP`, `(g\cdot f)(x) := g(f(g^{-1}x))`
Let `n := Card(P)`, then `\Gamma` is a `3n` dimensional vector space, which induces a `3n` dimensional representation `\rho_{dis}` of `G` with character `\chi_{dis}`.
-
We can show that this representation has two sub-representations consisting of global translations
and rotations of the molecule.
We calculate `\chi_{trans}` and `\chi_{rot}` the characters of these sub-representations, they account for 6 degrees of freedom. - Calculate `\chi_{vib} = \chi_{dis} - \chi_{trans} - \chi_{rot}`
-
Decompose the standard character on a basis of irreducible characters:
`\chi_0 = \sum_{i=1}^d \alpha_k \chi_k`
The theoretical number of bands on the IR spectrum is given by:`n_0 = \sum_{i=1, \alpha_i\ne0}^d \langle\chi_i, \chi_{vib}\rangle`
Example: IR Spectroscopy of Ammonia `NH_3`
We have the molecule of ammonia :
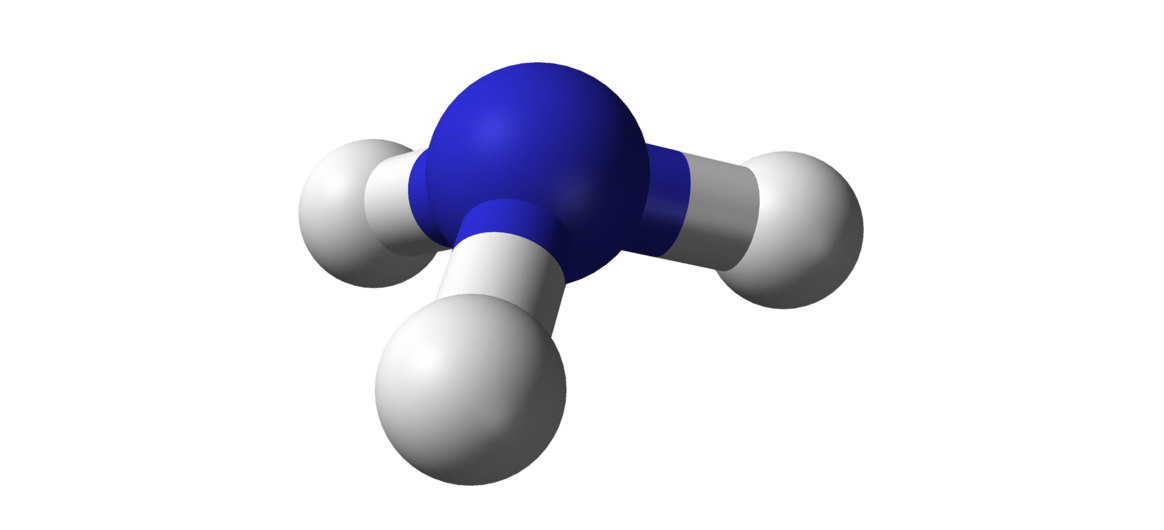
Let `G` be the symmetry group of this structure, the conjugacy classes of `G` are:
- `Id`
- `C_3` : 2 rotations of order 3
- `sigma` : 3 reflections of order 2.
| `D_3` | `Id` (1) |
`C_3` (2) |
`\sigma` (3) |
|---|---|---|---|
| `\chi_{triv}` | 1 | 1 | 1 |
| `\chi_1` | 1 | 1 | -1 |
| `\chi_2` | 2 | -1 | 0 |
Using the above method we compute the standard character (using the trace of rotation matrices: `tr(Id)=3, tr(C_3) = 0, tr(\sigma) = 1`), this gives:
| `D_3` | `Id` (1) |
`C_3` (2) |
`\sigma` (3) |
|---|---|---|---|
| `\chi_0` | 3 | 0 | 1 |
`\chi_0 = \chi_{triv} + \chi_2`
and`\chi_{dis} = 3\chi_{triv} + \chi_1 + 4\chi_2.`
The non vibration displacements give:`\chi_{trans} + \chi_{rot} = \chi_{triv} + \chi_1 + 2\chi_2.`
We obtain
`\chi_{vib} = 2\chi_{triv} + 2\chi_2.`
Thus:`n_0 = \langle\chi_{vib},\chi_{triv}\rangle + \langle\chi_{vib},\chi_{2}\rangle = 4.`
The analysis of the vibration modes of the ammonia molecule indeed shows four resonance frequencies respectively at:
`\nu_1 = 950` `cm^{-1}`
`\nu_1 = 1627` `cm^{-1}`
`\nu_1 = 3337` `cm^{-1}`
`\nu_1 = 3444` `cm^{-1}`
which you can visualize on this website, see [1].
Conclusion:
This article gives a basic computation method of certain absorption bands, however symmetries do not entirely determine the experimental IR spectrum of a molecule which is in practice much more complex and noisy.A slightly modified version of this method can also be used to determine the number of absorption bands in a Raman spectrum, see [2] for details.
Sources :
[1] Resonance data from NIST WebBook
[2] Group theory for the interpretation of IR & Raman Spectra
[3] Mathematical aspect and an other example with `PCl_5` (french)
[4] Molecular vibration and IR absorption (French)